Environmental Pollution and Control in Mines - MCQs from AMIE exams (Set 3)
1. An atmospheric radiative window exists between the wavelengths of
(a) 7 µm -12 µm
(b) 1 µm-3 µm
(c) 3 µm - 10 µm
(d) 20 µm-35 µm
2. ‘Scaled distance’ as used in prediction of PPV from ground vibrations, contains the following terms:
(a) charge per delay, frequency
(b) frequency, distance
(c) distance, charge per delay
(d) frequency, distance, charge per delay
3. Smog formation takes place due to
(a) nitrogen oxides.
(b) unburnt hydrocarbons.
(c) sunshine.
(d) All the three above.
4. Polyacrylamide is a
(a) flocculant.
(b) coagulant
(c) absorbent.
(d) drying agent
5. Nitrogen fixing bacteria are found with
(a) tubers.
(b) legumes.
(c) lemongrass.
(d) green leaves.
6. Ozone layer exists in
(a) ionosphere
(b) troposphere
(c) mesosphere
(d) stratosphere
7. If temperature inversion in the atmosphere commences from a level higher than the top of a chimney, the plume will then experience
(a) fanning
(b) looping
(c) fumigation
(d) recirculation
8. Danger limit of noise, as per DGMS norms, corresponds to
(a) 85 dBA
(b) 80 dBA
(c) 90 dBA
(d) 110 dBA
9. Dissolved oxygen level in fresh water may be of the order of
(a) 7 mg/1
(b) 2 mg/1
(c) 25 mg/1
(d) 72 mg/1
10. Turbidity of water is measured in the unit of
(a) COD
(b) NTU
(c) PPM
(d) BTU
Answers
1. (a) In the Earth's atmosphere this window is roughly the region between 8 and 14 μm, although it can be narrowed or closed at times and places of high humidity because of the strong absorption in the water vapour continuum or because of blocking by clouds.
2. (c) A ratio used to predict ground vibrations. As commonly used in blasting, scaled distance is the ratio of the distance from the blast, in metres, to the square root of the maximum instantaneous charge weight, in kilograms.
3. (d) It is the chemical reaction of sunlight, nitrogen oxides and volatile organic compounds in the atmosphere, which leaves airborne particles and ground-level ozone.
4. (a) Flocculating agents are chemical additives that cause suspended solids to form aggregates called flocs. These agents are used in water treatment, municipal and industrial waste treatment, mineral processing, and papermaking. Flocculating agents are either inorganic salts or water-soluble organic polymers.
5. (b) Nitrogen-fixing bacteria, microorganisms capable of transforming atmospheric nitrogen into fixed nitrogen. Plants that contribute to nitrogen fixation include the legume family – Fabaceae – with taxa such as clover, soybeans, alfalfa, lupins, peanuts, and rooibos. Legumes are able to form a symbiotic relationship with nitrogen-fixing soil bacteria called rhizobia. The result of this symbiosis is to form nodules on the plant root, within which the bacteria can convert atmospheric nitrogen into ammonia that can be used by the plant.
6. (d) The ozone layer is one layer of the stratosphere, the second layer of the Earth's atmosphere.
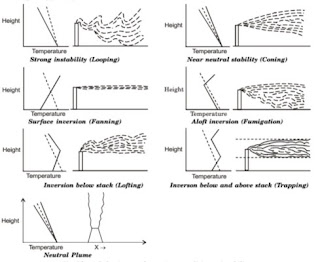
7. (c)
- Looping Plume: Occurs in super adiabatic environment. Unstable and need higher stacks.
- Neutral Plume: Upward vertical rise when ELR = ALR.
- Coning Plume: When wind velocity is high and sub-adiabatic conditions exist (ELR < ALR). The environment is highly stable.
- Fanning Plume: Under extreme inversion conditions. Emission will spread only horizontally.
- Lofting Plume: When there is strong super adiabatic condition above surface inversion. Such plume has minimum downward mixing, as its downward motion is prevented by inversion.
- Fumigating Plume: When inversion occurs above the top of the stack and super-adiabatic condition prevail below the stack.
- Trapping Plume: When inversion layer exist above the emission source as well as below the source. The emitted plume will neither go up nor go down.
8. (a) The operator and passengers shall not be exposed to noise level that exceeds an eight-hour equivalent continuous sound pressure level of 85 dB(A) and wherever it exceeds 85 dB(A), Personnel Protection Equipment (PPE) of adequate strength shall be used by the operators and passengers.
9. (a) Oxygen is considered as poorly soluble in water. Its solubility is related to pressure and temperature. In freshwater, DO reaches 14.6 mg/L at 0 °C and approximately 9.1, 8.3, and 7.0 mg/L at 20, 25, and 35 °C, respectively, and 1 atm pressure.
Dissolved oxygen (DO) is the amount of oxygen that is present in water. Water bodies receive oxygen from the atmosphere and from aquatic plants. Running water, such as that of a swift moving stream, dissolves more oxygen than the still water of a pond or lake.
10. (b) Turbidity is measured in NTU (Nephelometric Turbidity Units). The instrument used for measuring it is called nephelometer or turbidimeter, which measures the intensity of light scattered at 90 degrees as a beam of light passes through a water sample.
---
- The study material for AMIE/B Tech/Junior Engineer exams is available at https://amiestudycircle.com
- If you like the post please share your thoughts in the comment section

Comments